Buy sodium hypochlorite
While buying sodium hypochlorite from a reputable store with the best price, Dr. Chemical is at your service. You can prepare this solution with 14% active purity.

The price of sodium hypochlorite
What is sodium hypochlorite?
Jawal water is a chemical compound with the chemical formula NaClO. The sodium hypochlorite solution is the same as the bleaching liquid. Sodium hypochlorite is a white solid powder, but it is most commonly used in water-soluble form.
It is commonly called bleach, although what we know as household bleach contains small amounts of sodium chloride, sodium carbonate, and sodium hydroxide in addition to bleach to stabilize the product and maintain pH.
In general, water-soluble hypochlorite is used in different concentrations. Although solid acid hypochlorite is available, it is not used commercially. These solutions have greenish and yellowish liquids with chlorine smell. It is a white solid that easily decomposes into water releasing oxygen and chlorine. It also has a strong chlorine smell.
None of the compounds occur naturally in the environment. This solution is primarily used as a bleaching or disinfecting agent. Javel water is used as a commercial bleach, cleaning and disinfecting solution for drinking water and recycled water treatment systems and swimming pools.
Sodium hypochlorite is used in chemical pulps, textile bleach, commercial cleaner, household bleach, swimming pool disinfectant, municipal water and wastewater disinfectant, disinfectant in dairies and food processing plants, hospitals and households.
Hypochlorite is also used to control fungi in oil production equipment and as a sweetener (water softener) in oil refineries and in chemical industries to produce hydrazine, trisodium chloride phosphate and organic chemicals.
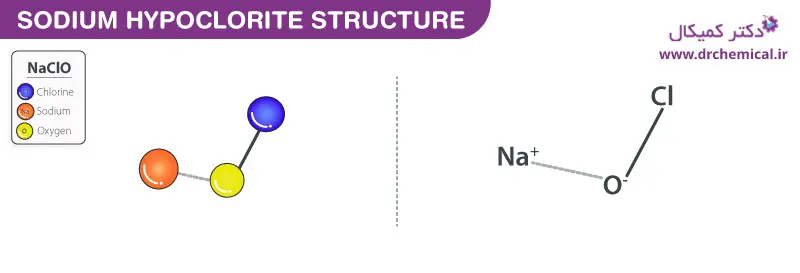
Structure of sodium hypochlorite
As it is clear from the chemical structure of Javal water, this compound consists of hypochlorite anion and sodium cation. Javal juice is available as a pale yellow to green dilute solution. It is an unstable, anhydrous compound that can decompose explosively. The average pH of this substance is 11.
Sodium hypochlorite has a sweet and chlorine-like smell. It is widely used as a cleaning or disinfecting agent and a bleaching agent. This chemical is widely used in many household detergent products.
Historically, bleaching liquid started in a simple process by passing an electric current through salt water to produce sodium hydroxide (NaOH) and chlorine gas (Cl2). Then, these two products are combined and produce Jawal Water Whitener.
Sodium hypochlorite solution
Sodium hypochlorite solutions, produced directly by electrolysis of seawater or salt water, are mainly used in wastewater and wastewater treatment , commercial disinfectants, large swimming pools and ships to inhibit bacterial growth in seawater systems, for the treatment of distilled water, and as a disinfectant for Fish stock is used. This solution is used to control sludge, shellfish and algae in plumbing and pipes, to disinfect seawater for secondary oil recovery and to remove cyanide.
Buying anti-algae with the best quality and market price

Properties of hypochlorite acid
Properties of sodium hypochlorite
| chemical formula | NaClO |
| Molecular weight | 74.44 g/mol |
| density | 1.11 grams per cubic centimeter |
| melting point | 18 degrees Celsius |
| boiling point | 101 degrees Celsius |
Dangers and side effects of sodium hypochlorite
Swallowing Perclin causes symptoms of stomach pain, burning sensation, cough, nausea, sore throat and vomiting. Direct contact with javel juice causes redness and discomfort in the skin or hair.
by adding javel water to water? How does the pH change
Due to the presence of caustic soda in hypochlorite, the pH of water increases. When it dissolves in water, two substances are formed that play a role in oxidation and disinfection. These are hypochlorous acid ( HOCl ) and the less active hypochlorite ion ( OCl -). The pH of the water determines the amount of hypochlorous acid.
While hypochlorite is used, hydrochloric acid ( HCl to lower the pH ) is used . Sulfuric acid ( H2SO4 ) can be used as a substitute for acetic acid. Less harmful gases are produced when using sulfuric acid. Sulfuric acid is a strong acid that reacts strongly with bases and is very corrosive.
Uses of Perclin
Sodium hypochlorite is widely used. For example, in agriculture, chemical industries, paint and lime industries, food industries, glass industries, paper industries, pharmaceutical industries, synthetic industries and waste disposal industries. In the textile industry, sodium hypochlorite is used to bleach textiles.
Sometimes it is added to industrial wastewater. This is done to reduce odors. sulfur ( SH ) and ammonia ( NH3 Hydrogen gas neutralizes ). It is also used to detoxify the cyanide bath in metal industries.
Hypochlorite can be used to prevent the growth of algae and oysters in cooling towers. In water purification, this solution is used to disinfect water. In households, hypochlorite is often used to purify and disinfect the house.
Salt water or sodium hypochlorite solution is one of the most versatile cleaning and disinfecting liquids available.
From doing basic household chores to disinfecting hospitals, from being used as a biocide to inhibit biological growth to deodorizing sewage, Perclin plays an effective role in a wide range of daily operations around the world.

Uses of Javal water
Buy biocide with quality assurance
- It is used as a key ingredient in laundry bleach. javel water hydrolyzes bonds, breaking up stains to make clothes easier to clean with detergent. It is for this reason that adding bleach along with detergent not only disinfects the clothes, but also makes them cleaner and whiter than regular washing.
- It is used as a bleaching agent in the paper and textile industries. Bleaching is the process of whitening wood pulp in order to improve printing properties and its ability to absorb liquids. Bleaching also attacks some contaminants to reduce stray dark dye particles in the final sheet of paper.
- Application for oxidation in different processes
- It is used in the refining of petroleum products.
- It is used in wastewater treatment.
- as an antiseptic . It is widely used
- It is used in food processing to disinfect food preparation equipment.
- It is used in swimming pools to prevent the growth of infectious agents.
How does Jawal water disinfection work?
By adding hypochlorite to water, hypochlorous acid ( HOCl ) is formed:
![]()
Hypochlorous acid is divided into hydrochloric acid (HCl) and oxygen (O). The oxygen atom is a very strong oxidizer. This solution is effective against bacteria, viruses and fungi. Sodium hypochlorite solution disinfects like chlorine.
How is Perclin used in swimming pools?
Hypochlorite is used in swimming pools for water disinfection and oxidation. It has the advantage that microorganisms cannot develop resistance against it. It is also effective against legionella bacteria and the biofilm in which legionella bacteria can multiply. Hypochlorous acid is produced by the reaction of sodium hydroxide with chlorine gas. So-called “active chlorine” is formed in water.
There are different ways to use this type of solution. For on-site salt electrolysis, salt solution ( NaCl ) in water is used. ) ions Sodium ( Na+ ) and chloride ( Cl- are produced.
![]()
By conducting the salt solution on an electrolysis cell, the following reactions take place in the electrodes:
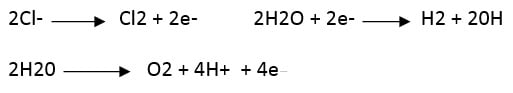
Subsequently, chlorine and hydroxide react to form hypochlorite:
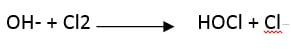
The advantage of the salt electrolysis system is that there is no need to transport or store hypochlorite. When this solution is stored for a long time, it becomes inactive. Another advantage of the site process is that chlorine lowers the pH and no other acid is needed to lower the pH.
The produced hydrogen gas contains explosives and as a result, ventilation is necessary to prevent leakage. This system is slow and additional sodium hypochlorite buffer needs to be used. Maintenance and purchase of electrolysis system is much more expensive than hypochlorite.
When this solution is used, acetic or sulfuric acid is added to the water. Excessive consumption can produce toxic gases. If the dose is too low, the pH rises and can irritate the eyes.
Since hypochlorite is used both to oxidize pollutants and to destroy pathogenic microorganisms, the required concentration of these solutions depends on the concentration of these pollutants. Especially, the amount of organic pollution determines the required concentration. If the water is filtered before applying sodium hypochlorite, calcium sodium deficiency is required.
Jawal water for swimming pool
The used concentration of this solution found in swimming pools is generally not harmful to people. When there is too much chlorine in water, it burns body tissues, causing damage to the respiratory system, stomach and intestines, eyes, and skin.
When sodium hypochlorite is used in swimming pools, it sometimes causes red eyes and a typical chlorine smell. When there is a large amount of urea (a mixture of urine and sweat), hypochlorous acid and urea react to form chloramine.
These chloramines irritate the mucous membranes and create the so-called chlorine smell. In most swimming pools, these problems are prevented by water purification and air conditioning. Eye irritation disappears after a while.

How sodium hypochlorite works
How to determine the concentration of sodium hypochlorite solution?
The concentration of hypochlorite depends on the characteristics of the local water. Usually, an amount in the range of 5-10 ml added to 20 liters of water is sufficient to inactivate disease-causing organisms, but not leave an unpleasant taste. Once the cap size for your project is determined, simple tests can be used to determine the proper concentration. To perform the tests, you need readily available sodium hypochlorite, source water in your area, and a kit that measures free and combined chlorine.
Industrial javel water
Brine is widely used in drinking water systems, swimming pools, etc., for chlorination. Chlorination usually produces small amounts of harmful byproducts. Javel water solutions have been used to treat dilute cyanide wastewaters such as electroplating wastes. The brine has also been used to treat more concentrated cyanide wastes, such as silver cyanide plating solutions. Toxic cyanide oxidizes to non-toxic cyanate (CNO-) and idealizes as follows:
CN- + ClO- >>> CNO- + Cl-
Javel water is commonly used as a biocide in industrial applications to control sludge and bacteria formation in water systems used in power plants, pulp and paper mills, etc. in solutions of 10-15% by weight.
Perclin is used in large quantities in various industries such as agriculture, chemical industries, paint and lime industries, food industries, glass industries, paper industries, pharmaceutical industries, synthetic industries and waste disposal industries.
Perclin is used in the textile industry to whiten textiles. This solution is sometimes added to industrial wastewater. This is done to reduce the smell.
Sodium hypochlorite neutralizes hydrogen sulphide (SH) and ammonia (NH3) gas. It is also used to detoxify cyanide bath in metal industry.
Bile water can be used to prevent algae and shellfish growth in cooling towers.

Javal water in water purification
In water purification, Javal water is used to disinfect water. In homes, Perclin is often used to purify and disinfect the home.
The concentration of water in the water reservoirs
If the water tank is new, and you know it is contaminated, perform a “shock chlorination” at 50-100 ppm and let it sit for 12-24 hours. After adding chlorine, the chlorine level starts to decrease. Chlorine is consumed and broken down depending on the chlorine requirement (your water chemistry and conditions) and water temperature.
Test the residual chlorine after 24 hours and repeat the procedure if the chlorine level is 10 PPM or less. If you are storing water and want to keep residual chlorine for safety, use a retention residual of 1 to 2 PPM.
Shock chlorination
In the shock method, chlorine concentration is added until the residual reaches 50 to 100 PPM. This is recommended when you have a new storage tank or well work has been done or you find that the storage tank is contaminated with coliform bacteria.
In shock chlorination, the water is rendered unfit for potable use until the chlorine level drops below 2 to 4 PPM, which occurs within days to weeks depending on water temperature and chemistry.
Low level maintenance or chlorination
If the water tank is for long-term storage or needs to be chlorinated periodically, the chart for adding 1 to 2 PPM is below:
- Step 1: First, clean the water storage tank. Clean the storage tank. Remove debris and hose off any dirt or other deposits or interior surfaces. If using pool chlorine (12% sodium hypochlorite), use half of the amount below.
- Step 2: Use a strong chlorine solution. If possible, clean the interior surfaces of the storage tank or tank with a strong chlorine solution containing ½ gallon of brine or ¼ gallon of pool chlorine per 5 gallons of water.
The following table is based on 5% Javal water. If you use pool chlorine (12% brine), use half of the values in the table:
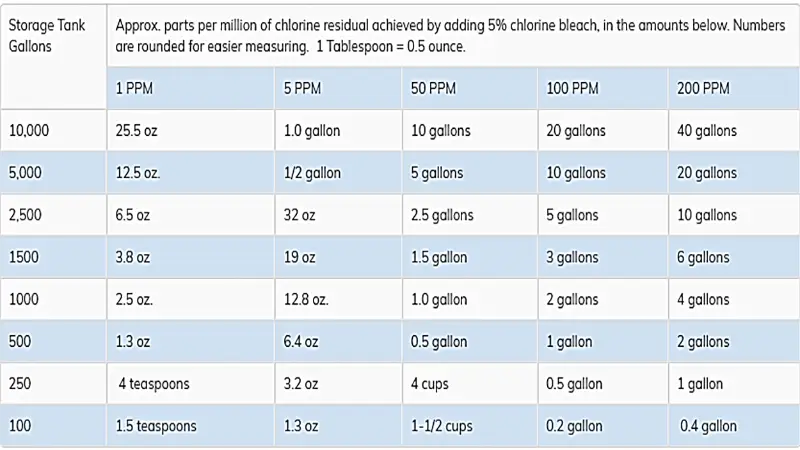
Jawal water concentration
The difference between Javal water and Vitex
Javal water is used for disinfection and washing at home, which is better known as Vitex.
Wholesale sale of juice
Sodium hypochlorite is used in many industries where quality is of great importance in various fields of industry. Buyers who need to buy and sell Javal water in bulk can contact Dr. Chemical store and receive the high quality product as soon as possible after inquiring about the price and receiving complete specifications.